The key to no findings is simple: finding the right partner. 
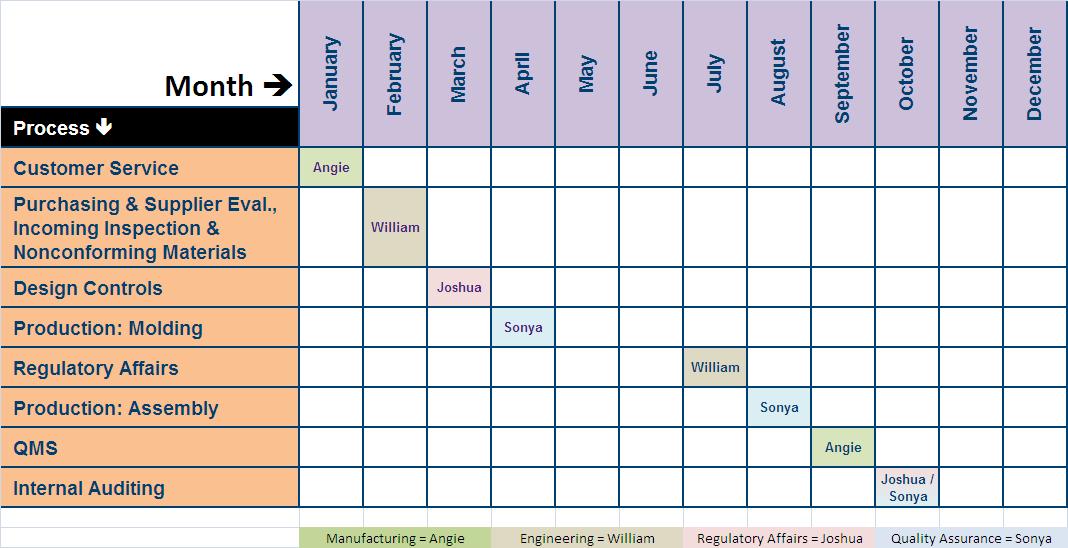
Internal audits are an ideal tool for consistently improving both your QMS and your compliance. We believe strongly in the effectiveness of the internal audits being a key indicator for the overall status of the QMS. RookQS’ team has completed over 100 remote internal audits, and can assist your team in completing your audit in the timeliest and most efficient manner. Rook’s Certified Auditors will conduct internal or supplier audits, and provide critical support during an external or regulatory audit, including FDA, MDSAP, ISO, and MDR. Installation, sterilization, and other manufacturing processes Example of Items Internal Audits Should Include: For the listing of information required for an ISO 13485:2016-compliant QMS, see this white paper: Checklist of. They will also check on the records and compliance with the procedures. While these are the documents and records that the EU MDR has identified as mandatory, it is important to note that this does not include any documented information necessary for the proper function of your Quality Management System. During this process, an auditor will check the compliance of the procedures to the selected regulatory regulations and standards. 
General Requirements EU Declaration of Conformity This is a formal document that officially certifies that your product fulfils the essential requirements needed to meet the applicable CE directives. 
What is an Internal Audit for a Medical Device?Īn internal Audit for medical devices is a process conducted by organizations that design or manufacture medical devices. For more information on how ISO 13485 relates to the CE marking, see: How to use ISO 13485 to get your devices approved for CE marking. RookQS has a seasoned team of 11 certified quality auditors (CQAs) for a variety of regulations, including FDA 21 CFR 820, ISO 13485:2016, MDSAP, and CE Mark under MDR. But many medical device companies struggle keeping up with the constant updates and new guidance documents for MDR, FDA, ISO, and other regulatory agencies. It addresses requirements for documentation, including quality manuals, operational procedures, specifications, and records. The ISO 13485:2016 / FDA-CFR Internal Audit Checklist This list has been prepared for you by the 13485 Store. Regulatory bodies worldwide require medical device companies to conduct internal audits. The internal audit checklist provides guidance on assessing compliance with ISO 13485 requirements for quality management systems. This document is an appendix to the main document, which is sold separately. The document is optimized for small and medium-sized organizations - we believe that overly complex and lengthy documents are just overkill for you. ISO 134 + Additional Requirements (and missing. The internal audit process is critical to emerging medical device companies to ensure that your Quality System is effective and compliant for the size and scope of your company. The Internal Audit Report is the document used to report on the findings of an internal audit. Management System sample editable documentation packages include Manual, procedures, SOPs, formats, audit checklist, etc. The checklist provides questions that refer to the ISO 13485: 2003 standard & US FDA requirements. When device makers are using the same language to communicate, there is less chance of miscommunication and misunderstandings that can lead to deviations and other quality events than cause patient harm and regulatory sanctions.It’s far better to identify any issues or gaps in your QMS during an internal audit than in front of the FDA or ISO. ISO 13485 compliance promotes harmonization, e.g., a standardized quality vocabulary. Increased credibility ultimately leads to increased brand equity, a desirable competitive advantage. Prospects recognize the value the certification and understand the investment in quality a company makes to earn it.Ĭompanies deemed ISO 13485 compliant receive a certificate which can be displayed on corporate advertising and marketing materials, leading to increased credibility with customers, employees and stakeholders. ISO 13485 certification establishes a company’s credibility and dedication to quality. Unclear or vague documentation, which is often what keeps company processes and ISO 13485 compliance at odds, is avoided. When company processes correspond to the ISO 13485 standard, it means they are transparent.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
